Generative AI in Healthcare 2026: 5 Use Cases Cutting Doc Time 50%

Key takeaways: Generative AI is entering production in healthcare with three primary use cases: automated clinical documentation reducing physician documentation time by over 50% using ambient clinical intelligence like Microsoft's DAX Copilot, diagnostic support through medical imaging analysis and patient record synthesis, and accelerated pharmaceutical research including candidate molecule generation and clinical trial optimization. The regulatory landscape requires triple compliance: AI Act high-risk obligations for medical AI systems by August 2026-2027, MDR medical device certification for class IIa and above, and GDPR Article 9 protections for sensitive health data requiring explicit consent or public interest justification. A hospital case study deploying AI documentation across 2,500 beds showed documentation time dropping from 15 to 7 minutes per patient, physician NPS rising from 12 to 47, and note completeness improving from 72% to 91%. Deployment methodology spans five phases: regulatory qualification over two to four months, clinical evaluation over three to six months, technical EHR integration, change management with clinician engagement from the design phase, and mandatory post-deployment surveillance. Ikasia offers healthcare AI audits, training for healthcare professionals, and medical AI project support.
The healthcare sector is experiencing a quiet revolution. Generative AI, long confined to POCs, is entering production in healthcare facilities: automated clinical documentation, diagnostic support, accelerated pharmaceutical research. But this highly regulated sector imposes specific constraints. How do you deploy generative AI in healthcare safely and compliantly?
Generative AI in Healthcare: 2026 Use Case Overview
Market Status
AI in healthcare represents a fast-growing market, estimated at tens of billions of dollars in 2025 with annual growth exceeding 30% according to market analysts.
Key segments (by market importance):
- Assisted diagnostics
- Clinical documentation
- Drug discovery
- Administration/management
- Patient monitoring
The 6 Major Use Cases for Generative AI in Healthcare
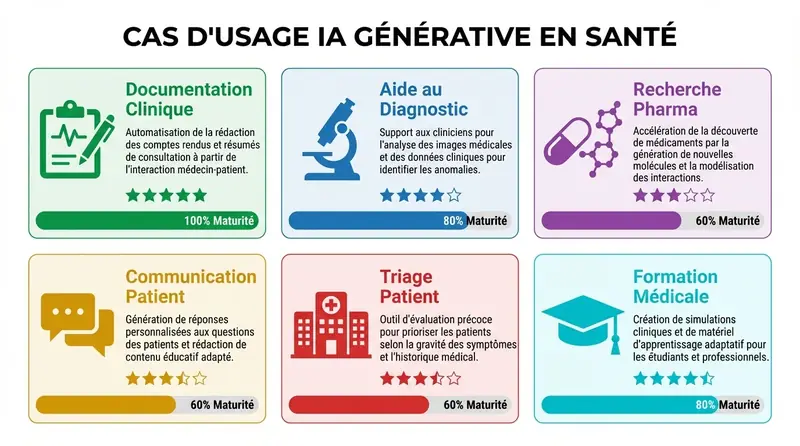
Clinical Documentation: Reducing Administrative Burden
The Problem
Physicians spend an average of 2 hours of documentation for every 1 hour of patient care. This administrative burden is a major cause of burnout and disengagement.
The Solution: Ambient Clinical Intelligence
Generative AI listens to the consultation (with patient consent) and automatically generates:
- Consultation summary
- Prescriptions for validation
- Diagnostic codes (ICD-10, CPT)
- Letter to referring physician
Example: DAX Copilot from Nuance/Microsoft
Deployed in several hospital systems since 2025:
Process:
1. Physician activates recording (patient consent)
2. Consultation proceeds normally
3. DAX transcribes and structures in real time
4. Physician validates/corrects the generated note
5. Automatic integration into EHR (Electronic Health Record)
Observed results:
- Documentation time: significant reduction
- Physician satisfaction: marked improvement
- Note completeness: notable improvement
- Coding errors: meaningful decrease
Points of Attention
- Patient consent: Mandatory and documented
- Human validation: The physician remains responsible
- EHR integration: Complex depending on vendors (Epic, Cerner, etc.)
- Language accuracy: Variable performance based on accents and jargon
Diagnostic Support: LLMs as Second Medical Opinion
The Concept
LLMs can analyze symptoms, patient history, and exams to suggest differential diagnoses. They don't replace the physician but enrich their thinking.
Use Case 1: Medical Imaging Analysis
Radiology (CT, MRI, X-ray):
- Anomaly detection (nodules, fractures, stroke)
- Emergency prioritization
- Automatic measurements (tumor size, volumes)
Dermatology:
- Skin lesion classification
- Early melanoma detection
- Evolution tracking
Ophthalmology:
- Diabetic retinopathy
- AMD
- Glaucoma
Use Case 2: Patient Record Synthesis
The LLM synthesizes the entire patient record to:
- Identify relevant medical history
- Detect drug interactions
- Alert to contraindications
Example medical prompt:
You are a medical assistant. Analyze this patient record and provide:
1. Summary of significant medical history
2. Current medications and potential interactions
3. Alerts on missing exams per clinical guidelines
4. Questions to ask the patient
Patient: [anonymized data]
Use Case 3: Differential Diagnosis
Typical workflow:
Symptoms + Exams + Medical History
↓
Medical LLM
↓
List of possible diagnoses
(with estimated probabilities)
↓
Physician evaluates and decides
Limitations and Precautions
What AI CANNOT do:
- Make a definitive diagnosis
- Prescribe treatment
- Replace physical examination
- Handle atypical cases
Identified risks:
- Automation bias: Over-reliance on AI
- Hallucinations: Made-up diagnoses
- Data bias: Under-representation of certain populations
- Liability: Who is responsible in case of error?
Pharmaceutical Research: Accelerating Discoveries
The Context
Developing a drug takes 10-15 years and costs an average of $2-3 billion. Generative AI can accelerate several stages.
Generative AI Applications in Pharma
1. Candidate Molecule Generation Generative models (diffusion models, GNNs) propose molecular structures optimized for a therapeutic target.
2. Toxicity Prediction Before synthesis, AI predicts potential toxicity and filters out dangerous candidates.
3. Clinical Trial Optimization
- Patient selection (inclusion criteria)
- Dropout prediction
- Side effect detection
4. Scientific Literature Analysis LLMs analyze millions of articles to identify:
- New therapeutic targets
- Repurposing of existing drugs
- Research trends
Example: AlphaFold and Structural Biology
DeepMind's AlphaFold has revolutionized protein structure prediction:
- Database of 200 million predicted structures
- Considerable time savings for certain research stages
- Applications in drug development, agriculture, environment
AI Act Requirements for Medical AI Devices
Classification of Healthcare AI Systems
The AI Act classifies medical AI systems primarily as high-risk:
High-risk systems (Annex III):
- Medical devices (class IIa and above under MDR)
- Diagnostic support systems
- Systems influencing treatment decisions
- Emergency triage tools
Specific obligations:
| Obligation | Detail |
|---|---|
| Risk management system | Identification, assessment, mitigation of risks |
| Data governance | Quality, representativeness, traceability of datasets |
| Technical documentation | Complete system description |
| Transparency | Information to users about capabilities/limitations |
| Human oversight | Possibility of human override |
| Accuracy and robustness | Documented performance testing |
| Cybersecurity | Protection against attacks |
AI Act / MDR Articulation
The European Medical Device Regulation (MDR 2017/745) applies IN ADDITION to the AI Act:
A medical AI system is subject to three simultaneous regulatory frameworks:
- AI Act (if AI) -- High-risk obligations
- MDR (if medical device) -- CE marking, clinical evaluation
- GDPR (if personal data) -- Legal basis, patient rights
Compliance Timeline
| Date | Obligation |
|---|---|
| February 2025 | Prohibited practices + AI literacy |
| August 2025 | GPAI obligations |
| August 2026 | Full high-risk obligations |
| August 2027 | Medical device AI (class IIa+) |
GDPR and Health Data: The Specific Legal Framework
Health Data is Sensitive Data
GDPR (Article 9) prohibits in principle the processing of health data, except for exceptions:
- Explicit patient consent
- Necessity for care
- Public interest in public health
- Scientific research (with safeguards)
Legal Basis for Medical AI
In care context:
- Legal basis: Public interest mission + necessity for care
- No consent required for care itself
- BUT clear patient information about AI use
In research context:
- Legal basis: Public interest in scientific research
- Ethics committee approval required
- Pseudonymization or anonymization necessary
Patient Rights
| Right | Medical AI Application |
|---|---|
| Information | Know that AI is being used |
| Access | Obtain data used |
| Rectification | Correct errors |
| Objection | Refuse AI use for their care |
| Explanation | Understand AI decision logic |
| Human intervention | Request a human decision |
Focus: Health Data Platforms
Health data platforms (like NHS Digital, French Health Data Hub) centralize health data for research:
- Controlled access for research projects
- Regulatory authorization procedures
- Secure analysis environment
Use for AI:
- Model training on real data
- Validation on national cohorts
- National performance benchmarking
Deploying AI in Healthcare: Methodology
Phase 1: Regulatory Qualification (2-4 months)
Key questions:
- Is the tool a medical device under MDR?
- What AI Act risk class?
- What data is processed?
Deliverables:
- Documented regulatory analysis
- Compliance strategy
- Budget and timeline
Phase 2: Clinical Evaluation (3-6 months)
Objectives:
- Demonstrate clinical safety
- Measure real-world performance
- Identify biases and limitations
Method:
1. Define performance criteria (sensitivity, specificity, etc.)
2. Constitute representative test dataset
3. Prospective evaluation on real cases
4. Error analysis and adjustments
5. Complete documentation
Phase 3: Technical Integration (2-4 months)
Points of attention:
- EHR interoperability (HL7 FHIR, IHE)
- Strong authentication (healthcare credentials)
- Health data hosting compliance
- Access traceability
Phase 4: Change Management (ongoing)
Barriers on the clinician side:
- Distrust of technology
- Fear of replacement
- Perceived workload for transition
Adoption levers:
- Involve clinicians from design phase
- Train on concrete benefits (time savings)
- Reassure about human validation role
- Measure and communicate results
Phase 5: Post-Deployment Surveillance
MDR/AI Act obligations:
- Continuous performance monitoring
- Incident reporting
- Regular system updates
- Periodic audit
Case Study: AI Documentation Deployment at Hospital
Context
A 2,500-bed hospital system wants to deploy AI clinical documentation.
Approach
Months 1-2: Scoping
- Regulatory audit: Class I medical device
- Solution choice: DAX Copilot (Microsoft/Nuance)
- Project team formation (physicians, IT, DPO, legal)
Months 3-4: Pilot
- 20 volunteer physicians (3 departments)
- Initial training (4h)
- Continuous quality/satisfaction evaluation
Months 5-6: Evaluation
- Analysis of generated notes (quality, completeness)
- Time saved measurement
- User feedback
- Workflow adjustments
Months 7-12: Progressive Deployment
- Extension in waves (50 → 150 → 500 physicians)
- Internal trainer development
- Institutional communication
Results
| Metric | Before | After | Change |
|---|---|---|---|
| Documentation time/patient | 15 min | 7 min | -53% |
| Physician satisfaction (NPS) | 12 | 47 | +35 pts |
| Note completeness | 72% | 91% | +19 pts |
| Coding errors | 8% | 5% | -3 pts |
Our Healthcare AI Support
At Ikasia, we support healthcare organizations:
Healthcare AI Audit (3 days)
- Mapping of relevant use cases
- Regulatory analysis (MDR, AI Act, GDPR)
- Deployment roadmap
"AI for Healthcare Professionals" Training (1 day)
- Understanding AI capabilities and limitations
- Medical practical cases
- Ethical and regulatory issues
Medical AI Project Support (3-12 months)
- Complete project management
- Regulatory compliance
- Change management
Conclusion
Generative AI is tangibly transforming healthcare in 2026: automated clinical documentation, diagnostic support, accelerated research. But this highly regulated sector requires a rigorous approach.
Keys to success:
- Start with clinical documentation: Quick ROI, controlled risk
- Anticipate compliance: AI Act + MDR + GDPR = regulatory complexity
- Involve clinicians: Adoption depends on their engagement
- Keep humans at the center: AI assists, the physician decides
The potential is immense: reducing administrative burden, improving diagnostic quality, accelerating therapeutic discoveries. But only organizations that master both technology AND regulation will reap the benefits.
Enjoyed this article? Check out our Generative AI Workshop — 3.5h hands-on session to master the tool with your team.
Tags
Related courses
Related articles

AI Compliance for European Enterprises: Beyond the AI Act — A Practical Framework
Read
DeepMind's AI Co-Clinician: What the Medical Revolution Means for Every French Company
Read
GPT-5 and Unpredictable Behaviors: What the 'Goblins' Incident Reveals About AI Risks in Business
ReadWant to go further?
Ikasia offers AI training designed for professionals. From strategy to hands-on technical workshops.